|
A prokaryotic expression vector pET 28 a-His-SUMO-3 A(1-61) expressing the solution region of EV-D68 3 A was successfully constructed and plenty of highly pure target proteins were obtained by multiple purification steps. Chemical cross-linking reaction assay was taken to determine the multiple polymerization state of the 3 A soluble region. After that, the target protein 3 A(1-61) was purified by a series of purification methods such as Ni-NTA, anion exchange chromatography and gel filtration chromato- graphy. The recombinant protein was purified by Ni-NTA Agarose and cleaved by ULP Protease to remove His-SUMO tag. The recombinant plasmid was transformed into Escherichia coli BL21 induced by IPTG to express the fusion protein His-SUMO-3 A(1-61). The EV-D68 3 A(1-61) gene was amplified by PCR and then cloned into the expression vector pET- 28 a-His-SUMO. To understand the structure of the soluble region of Enterovirus 68 3 A protein, we construct a prokaryotic expression vector expressing the soluble region of EV-D68 3 A protein, and identify the forms of expression product after purification. Li, Ting Kong, Jia Yu, Xiao-fang Han, Xue The antigenicity of the purified survivin protein was demonstrated desirable. 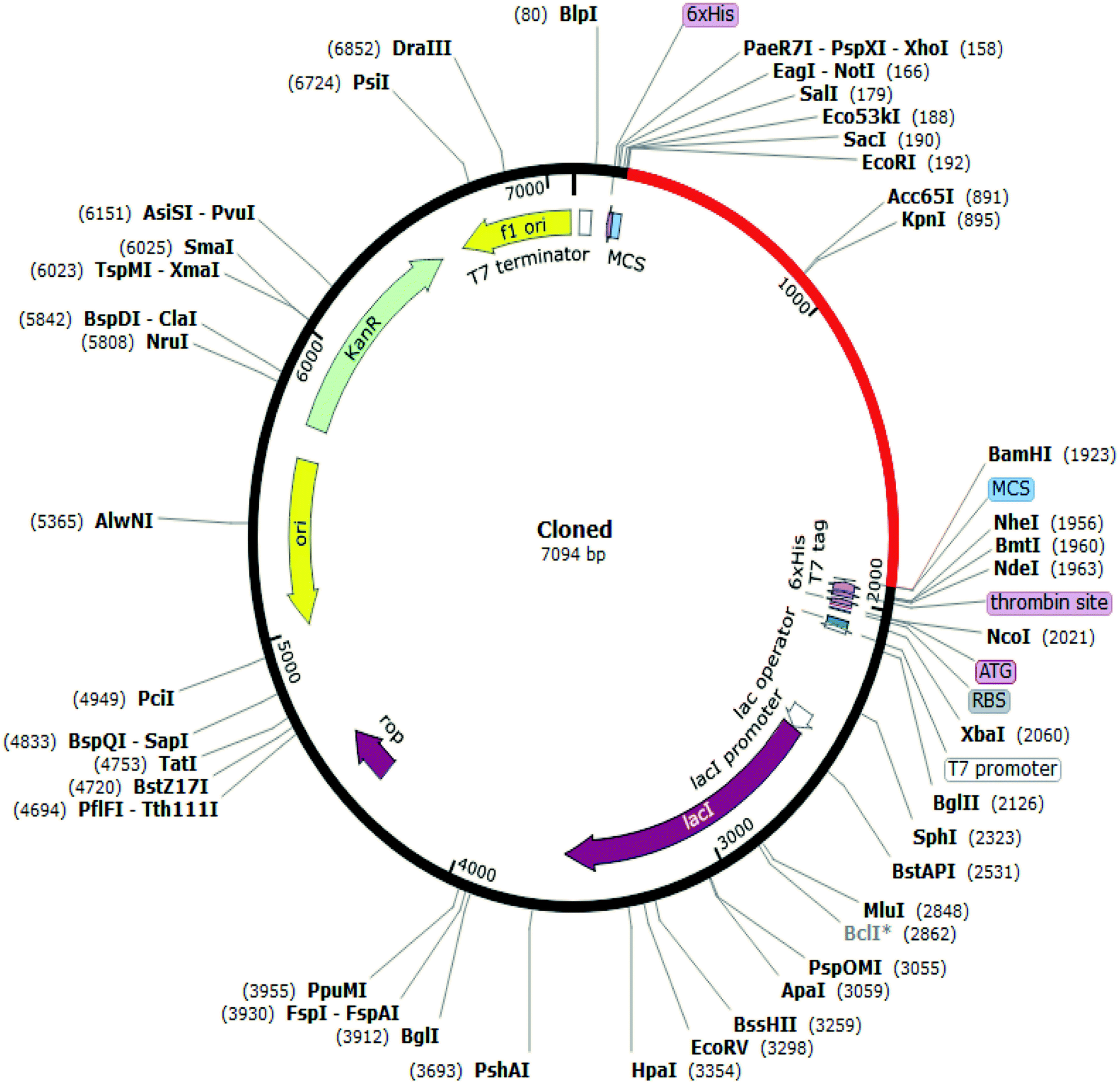
The prokaryotic expression plasmid pET 28 a-survivin was successfully constructed and the survivin protein was expressed and purified in E.coli. And the antigenicity of the survivin protein was validated by Western blotting and ELISA. The purity of the purified protein reached 90% by SDS-PAGE analysis. The fusion protein induced by IPTG was obtained with Mr about 24 000. The recombinant expression vector was verified successfully by BamHI and HindIII. The antigenicity of the purified survivin protein was identified by Western blotting and ELISA. The fusion protein was purified through Ni affinity chromatography. The expression vector was transformed into BL21 (DE3) and the fusion protein survivin/His was induced by IPTG. Survivin cDNA segment was amplified by PCR and cloned into prokaryotic expression vector pET 28 a(+) to construct the recombinant expression vector pET 28 a-survivin. To construct a prokaryotic expression plasmid pET 28 a-survivin, optimize the recombinant protein expression conditions in E.coli, and purify the survivin recombinant protein and identify its antigenicity. Yin, Xiaotao Wang, Wei Tian, Renli Xu, Yuanji Yan, Jinqi Zhang, Wei Gao, Jiangping Yu, Jiyun The method reported to accomplish plasmid reconstruction is uniquely versatile yet simple, relying on the strategic placement of primers combined with homologous recombination of PCR products in yeast. 
We also establish that progressively cooling the vector and insert leads to a significant increase in ligation-independent transformation efficiency, demonstrated by the incorporation of a 10.3 kb insert into the vector. This cassette contains dual inverted restriction sites that reduce false positive clones by generating a linearized plasmid incapable of self-annealing after a single restriction-enzyme digest. To address this issue, a method was developed to quickly exchange the multiple-cloning region of the popular expression plasmid pET- 28 with a ligation-independent cloning cassette, generating pGAY- 28. Unfortunately, it is common practice to amend or neglect protein targets if the gene that encodes the protein of interest is incompatible with the multiple-cloning region of a preferred expression vector. The ability to rapidly customize an expression vector of choice is a valuable tool for any researcher involved in high-throughput molecular cloning for protein overexpression. Rapid modification of the pET- 28 expression vector for ligation independent cloning using homologous recombination in Saccharomyces cerevisiae
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
